Test to detect H 2 gas: When burning matchstick is kept on the mouth of this test tube, pop sound is heard which confirms the presence of H 2 gas. The evolution of gas is confirmed by the bubble formation in soap solution. to liberate hydrogen gas and corresponding salt. It is observed that active metals like zinc react with strong bases like NaOH, KOH etc. Name the gas which will be evolved when the same metal reacts with dilute solution of a strong acid. Write the equation of the chemical reaction involved and the test to detect the gas. When the content are warmed, a gas evolves which is bubbled through a soap solution before testing. (ii) If CO 2 is passed for long duration through lime water, the white precipitate formed dissolves due to the formation of soluble calcium hydrogen carbonate and the solution becomes clear.Ģ mL of sodium hydroxide solution is added to a few pieces of granulated zinc metal taken in a test tube. (i) When CO 2 is passed through lime water for short interval of time, it turns milky due to the formation of insoluble calcium carbonate. (ii) for a long duration? Also write the chemical equations for the reactions involved. What is observed when carbon dioxide gas is passed through lime water Hence, oxides of non-metals are acidic in nature. Calcium hydroxide, which is a base, reacts with carbon dioxide to produce salt and water. For example, the reaction between carbon dioxide and calcium hydroxide. Oxides of non-metals react with bases to form salt and water.

What do you infer about the nature of non-metal oxide? (Board Term I, 2017) With the help of an example explain what happens when a base reacts with a non- metallic oxide. Important Questions of Acids Bases and Salts Class 10 Science Chapter 2 Previous Year Questions & Important Questions of Acids Bases and Salts Class 10 Science Chapter 2 will help the students to score good marks in the board examination. We have given these Important Questions for Class 10 Science Chapter 2 Acids Bases and Salts to solve different types of questions in the exam. Lakhmir Singh Science Class 8 Solutions.PS Verma and VK Agarwal Biology Class 9 Solutions.NCERT Solutions for Class 9 Foundation of IT.NCERT Solutions for Class 9 Social Science.NCERT Solutions for Class 10 Foundation of Information Technology.NCERT Solutions For Class 10 Hindi Kritika.NCERT Solutions For Class 10 Hindi Kshitiz.NCERT Solutions For Class 10 Hindi Sparsh.NCERT Solutions For Class 10 Hindi Sanchayan.NCERT Solutions for Class 10 Social Science.NCERT Solutions for Class 11 Indian Economic Development.NCERT Solutions for Class 11 Political Science.NCERT Solutions for Class 11 Psychology.NCERT Solutions for Class 11 Entrepreneurship.NCERT Solutions for Class 11 Accountancy.NCERT Solutions for Class 11 Business Studies.NCERT Solutions for Class 11 Computer Science (Python).NCERT Solutions for Class 12 Psychology.NCERT Solutions for Class 12 Political Science.NCERT Solutions for Class 12 Entrepreneurship.
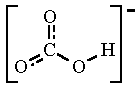
NCERT Solutions for Class 12 Macro Economics.NCERT Solutions for Class 12 Micro Economics.NCERT Solutions for Class 12 Accountancy.NCERT Solutions for Class 12 Business Studies.

SODIUM HYDROGEN CARBONATE FORMULA PDF
